| 0.018″ | 球囊长度 (mm) | |||
| 球囊直径 (mm) | 20mm | 40mm | 60mm | 80mm |
| 4.00mm | √ | √ | √ | √ |
| 5.00mm | √ | √ | √ | √ |
| 6.00mm | √ | √ | √ | √ |
| 7.00mm | √ | √ | √ | √ |
| 8.00mm | √ | √ | √ | √ |
| 0.035″ | 球囊长度 (mm) | |||
| 球囊直径 (mm) | 20mm | 40mm | 60mm | 80mm |
| 4.00mm | √ | √ | √ | √ |
| 5.00mm | √ | √ | √ | √ |
| 6.00mm | √ | √ | √ | √ |
| 7.00mm | √ | √ | √ | √ |
| 8.00mm | √ | √ | √ | √ |

用于扩张髂动脉、股动脉、髂股动脉、腘动脉、腘下动脉和肾动脉内的病变,治疗自体或人工透析动静脉内瘘阻塞。
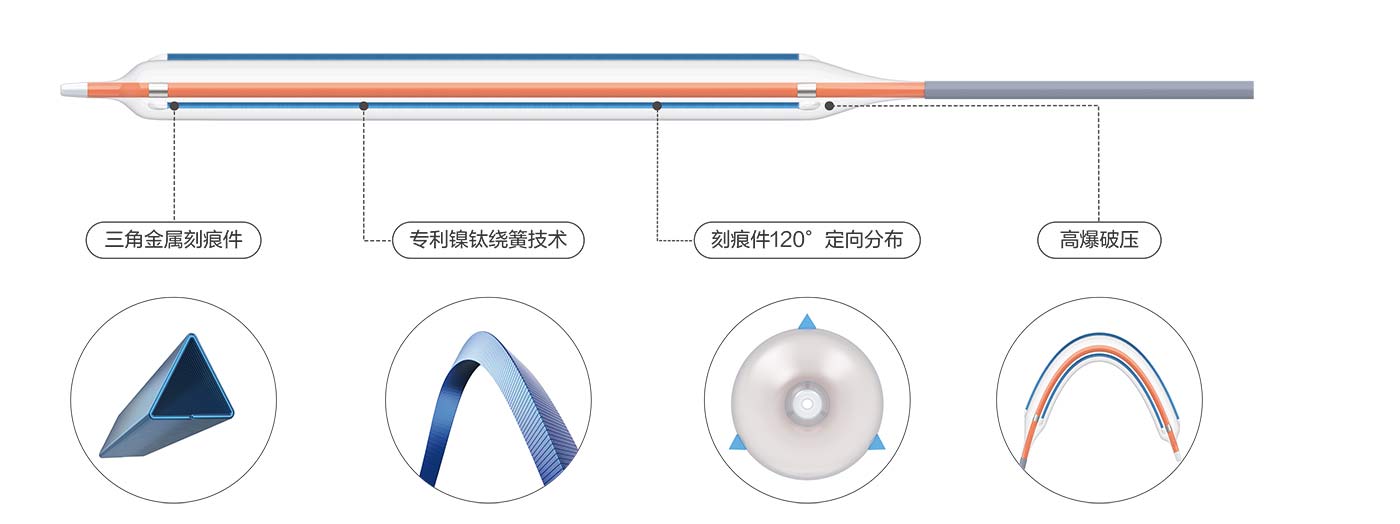
技术规范
| 适配导丝 | 0.018″/0.035″ |
| 球囊标称压力(atm) | 8 atm |
| 额定爆破压力(atm) | 20 atm |
| 球囊材料 | Nylon12 |
| 球囊标称直径(mm) | 4.0/5.0/6.0/7.0/8.0 mm |
| 球囊标称长度(mm) | 20/40/60/80 mm |
| 适配导管鞘 | DKT18: 6Fr(4.0mm, 5.0mm, 6.0mm)
DKT18: 7Fr(7.0mm, 8.0mm) DKT35: 6Fr(4.0mm, 5.0mm) DKT35: 7Fr(6.0mm, 7.0mm, 8.0mm) |
| 导管有效长度(cm) | 50/90/130 cm |
| 类型 | 整体交换型球囊扩张导管 |
| 刻痕件 | 镍钛合金 |
| 刻痕件高度(mm) | 0.36 mm |
球囊结构

| 0.018″ | 球囊长度 (mm) | |||
| 球囊直径 (mm) | 20mm | 40mm | 60mm | 80mm |
| 4.00mm | √ | √ | √ | √ |
| 5.00mm | √ | √ | √ | √ |
| 6.00mm | √ | √ | √ | √ |
| 7.00mm | √ | √ | √ | √ |
| 8.00mm | √ | √ | √ | √ |
| 0.035″ | 球囊长度 (mm) | |||
| 球囊直径 (mm) | 20mm | 40mm | 60mm | 80mm |
| 4.00mm | √ | √ | √ | √ |
| 5.00mm | √ | √ | √ | √ |
| 6.00mm | √ | √ | √ | √ |
| 7.00mm | √ | √ | √ | √ |
| 8.00mm | √ | √ | √ | √ |
技术规范
| 适配导丝 | 0.018″/0.035″ |
| 球囊标称压力(atm) | 8 atm |
| 额定爆破压力(atm) | 20 atm |
| 球囊材料 | Nylon12 |
| 球囊标称直径(mm) | 4.0/5.0/6.0/7.0/8.0 mm |
| 球囊标称长度(mm) | 20/40/60/80 mm |
| 适配导管鞘 | DKT18: 6Fr(4.0mm, 5.0mm, 6.0mm)
DKT18: 7Fr(7.0mm, 8.0mm) DKT35: 6Fr(4.0mm, 5.0mm) DKT35: 7Fr(6.0mm, 7.0mm, 8.0mm) |
| 导管有效长度(cm) | 50/90/130 cm |
| 类型 | 整体交换型球囊扩张导管 |
| 刻痕件 | 镍钛合金 |
| 刻痕件高度(mm) | 0.36 mm |
球囊结构

